Review the 2021 IPPS Proposed Rule’s MS-DRG & Other ICD-10 Coding Updates
Review the 2021 IPPS Proposed Rule’s MS-DRG & Other ICD-10 Coding Updates
CMS released the 2021 Proposed Rule for Inpatient Prospective Payment System (IPPS) on May 11. CMS published this in the May 29, 2020, Federal Register (2020). Provisions in the rule increase the Medicare IPPS rate for hospitals that report quality data and are meaningful users of EHRs by approximately 3.1%, resulting in an increase of $2.07 billion in spending. With other adjustments, the overall payment increase for inpatient hospitals will be around 1.6%. Additional updates include ICD-10-CM/PCS code additions/revisions/deletions, MCC/CC additions/revisions/deletions, DRG changes/revisions, and continued/discontinued New Technology Add-On payments. The 2021 IPPS Final Rule is now live – review it here.
Need to catch up on the ICD-10-CM, ICD-10-PCS, and IPPS & MS-DRG updates? Enroll in our ICD-10 Updates courses!
Adjustments to payment rates
The Proposed Rule for 2021 ICD-10 codes adjusts the payment rates, MS-DRGs, and other provisions. Among the changes is a proposal for a new MS-DRG code for Chimeric Antigen Receptor (CAR) T-Cell Therapies. Current hospital rates for CAR-T cases are paid at the same rate as bone marrow transplants, with additional payments via the new technology add-on payments. However, these add-on payments expire in FY 2021. CMS also proposed other new MS-DRGs, as well as changes in both diagnosis and procedure codes that group to MS-DRGs. CMS is also proposing changes for the new technology add-on payment (NTAP) for certain antimicrobial products that meet the alternative pathway criteria. This would allow these add-on payments to take effect before FDA approval.
New ICD-10 codes
The Proposed Rule also includes new ICD-10-CM and ICD-10-PCS codes. Though CMS published the codes in the proposed and final rule, they are not subject to comment. The majority of the more than 400 new diagnosis codes include expansions of categories and subcategories. There are new codes for sickle cell and other types of anemia and cytokine release, a syndrome that sometimes occurs after CAR-T therapy. This syndrome has also recently been associated with COVID-19 cases.
Substance use changes
Other changes include expanded substance abuse codes involving withdrawal, corneal dystrophies, and complications of corneal transplants. The Proposed Rule includes specific codes for synthetic narcotics fentanyl and tramadol poisoning. This also includes adverse effects and underdosing of these and other synthetic narcotics. These more specific codes will supply data for the fight against opioid addiction. There are additions to the External Cause codes, particularly for scooter accidents. The Proposed Rule also includes the category U07 codes for Vaping-related disorder and COVID-19 that were effective on April 1, 2020. See Table 6A appended to the Proposed Rule for a complete list of the proposed ICD-10-CM diagnosis codes (CMS, 2020).
Changes to ICD-10-PCS
There are more than 200 new ICD-10-PCS codes that include Fragmentation procedures in the heart and great vessels, and new code tables for Fragmentation of the upper and lower arteries and veins, with the addition of a qualifier for ultrasonic. CMS added new approach values for via natural or artificial opening and via natural or artificial opening endoscopic for Drainage of the pelvic cavity in code table 0W9. The agency also added a new Measurement code for musculoskeletal compartment pressure. Additionally, CMS added a new code table, BF5, for Other imaging in the Hepatobiliary System and Pancreas. CMS also added many new codes in the Radiation Therapy section for Brachytherapy utilizing the Cesium-131 isotope. Finally, the Proposed Rule includes six new Section X codes, all for Introduction of specified substances. See Table 6B for a full list of the latest ICD-10-PCS codes (CMS, 2020).
There are lists of additions and deletions to the CC exclusion, CC, and MCC lists due to the code updates. The 12 new ICD-10-PCS codes for inpatient COVID-19 admissions and treatments would also be included.
Here are a few of the changes that CMS has proposed for 2021:
Changes to MS-DRG
Creation of MS-DRG 521 and 522. One potential new change involves creating new MS-DRGs 521 and 522 for hip replacements that are performed with a diagnosis of a hip fracture. The hip fracture can be from trauma, pathologic, osteoporosis, stress, displaced or nondisplaced.
Revision of MS-DRG 037, 038, and 039. This potential change involves MS-DRGs 037, 038, 039 (Extracranial Procedures with MCC, with CC and without CC/MCC respectively) which are for carotid artery stent procedures. These DRGs all involve the dilatation of a carotid artery (common, internal or external) with intraluminal device(s).

If the proposed rule as approved, CMS will move the 6 procedures above to MS-DRGs “034, 035 and 036 (Carotid Artery Stent Procedures with MCC, with CC and without CC/MCC respectively)” (Federal Register, 2020).
Reclassifications and Revisions
Reclassification of Bone Marrow Transplants. Currently, MS-DRG 014 allogeneic bone marrow transplant, MS-DRG 016 autologous bone marrow transplant with CC/MCC or T-Cell Immunotherapy, MS-DRG 017 autologous bone marrow transplant without CC/MCC are currently designated as surgical MS-DRG’s. The proposal is to reclassify these DRGs to medical DRGs, since “bone marrow transplants involve a transfusion of donor cells and do not involve a surgical procedure or require the resources of an operating room” (Federal Register, 2020).
Revision of Left Atrial Appendage Closure (LAAC). Additionally, a proposal was made to create new MS-DRG specific to all left atrial appendage closure procedures or to reassign ICD-10-PCS procedure code 02L73DK from MS-DRG 274 to MS-DRG 273. The table below describes the proposed code assignments for LAAC procedures.

Miscellaneous
Addition of 9 ICD-10-PCS Codes for Totally Implantable Vascular Access Devices (TIVADs). The Proposed Rule wants to add 9 ICD-10-PCS procedure codes describing TIVADs as positioned in the table below to the MS-DRGs that document ‘‘Other’’ procedures within each of MDCs 04, 06, 07, 08, 13, and 16.

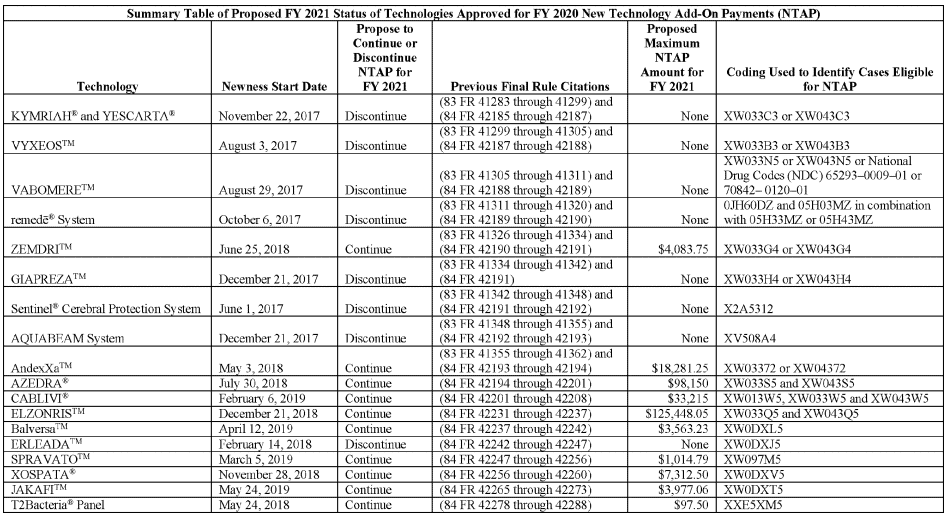
Revisions to the New Technology Add-On Payment Status of Current New Technologies as of FY2020. Review the table below to see which technologies the Proposed Rule suggests for discontinuation and continuation.
The IPPS Proposed Rule includes additional information on various issues. This includes building on price transparency by publishing hospital rates by MS-DRGs, inpatient quality reporting program, and graduate education funding. See the full 1600-page Proposed Rule for more details (Federal Register, 2020). CMS is no longer accepting comments on the IPPS Proposed Rule, which closed on July 10. 2020. The IPPS Final Rule went into effect on October 1, 2020. Navigate to the YES Blog for additional updates on the Inpatient Prospective Payment System Final Rule.
Additional Information
You can find further information on these guidelines and a comprehensive review of all of the changes for IPPS and MS-DRG in the ICD-10 Updates collection by YES HIM Education. It covers the applicable guidelines and identify new and/or revised information. The course will also analyze additions and deletions to the MCC and CC lists. The course also includes changes to add-on payments.
Carol Owen, Senior Coding Consultant at YES, co-authored this article with Teri Jorwic, MPH, RHIA, CCS, CCS-P, FAHIMA.